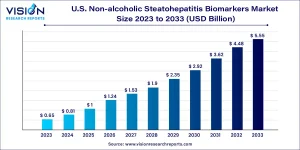
The U.S. non-alcoholic steatohepatitis (NASH) biomarkers market was valued at USD 0.81billion in 2024 and is expected to reach USD 5.55 billion by 2033, advancing at a CAGR of 23.93% between 2024 and 2033.
The U.S. NASH biomarkers market is shaped by a combination of drivers, opportunities, and challenges that are influencing its rapid evolution. Among the key drivers, the rising prevalence of lifestyle-related diseases such as obesity, type 2 diabetes, and metabolic syndrome has significantly increased demand for reliable diagnostic tools. At the same time, healthcare systems are placing greater emphasis on preventive care, making early detection through biomarkers a critical strategy for reducing long-term treatment costs.
Introduction: Understanding NASH and Its Growing Burden
Non-alcoholic steatohepatitis (NASH) is a progressive and severe form of non-alcoholic fatty liver disease (NAFLD), characterized by the accumulation of fat in the liver accompanied by inflammation and liver cell injury. Unlike alcohol-related liver conditions, NASH develops in individuals who consume little to no alcohol, making it a hidden yet significant health challenge. In the United States, rising rates of obesity, type 2 diabetes, and sedentary lifestyles have fueled the prevalence of NASH, positioning it as one of the fastest-growing causes of chronic liver disease. Alarmingly, many patients remain undiagnosed until the disease reaches advanced stages, underscoring the urgent need for early detection strategies. Biomarkers, offering non-invasive and reliable diagnostic insights, are emerging as powerful tools to address this unmet need and reshape the future of liver disease management.
U.S. Non-alcoholic Steatohepatitis Biomarkers Market Overview
Non-alcoholic steatohepatitis (NASH) is a progressive liver disease characterized by fat buildup, inflammation, and liver cell damage. With the rising incidence of obesity, diabetes, and metabolic syndrome in the United States, the demand for accurate and early detection methods has surged. Biomarkers play a crucial role in diagnosing, monitoring, and managing NASH, as they provide non-invasive alternatives to liver biopsies and support drug development. The U.S. market is evolving rapidly, fueled by ongoing research, strategic collaborations, and a strong pipeline of diagnostic and therapeutic innovations.
Access Your Sample Report Today: https://www.visionresearchreports.com/report/sample/41348
U.S. Non-alcoholic Steatohepatitis Biomarkers Market Growth
The U.S. NASH biomarkers market is experiencing significant expansion due to heightened awareness and advancements in precision medicine. Pharmaceutical companies, diagnostic firms, and research organizations are investing heavily in biomarker discovery to improve early detection and enhance treatment monitoring.
Additionally, the growing clinical trials landscape, supported by regulatory initiatives, is expected to accelerate adoption. As non-invasive diagnostic solutions gain traction over traditional methods, the market is poised for sustained growth in the coming years.
U.S. Non-alcoholic Steatohepatitis Biomarkers Market Trends
- Shift Towards Non-invasive Diagnostics: Traditional liver biopsies are invasive, costly, and carry risks for patients. As a result, the market is shifting toward blood-based, imaging, and genetic biomarkers that offer safer, faster, and more patient-friendly alternatives. These innovations are not only improving clinical adoption but also increasing compliance among patients who previously avoided diagnostic procedures.
- Increased R&D Collaboration: The U.S. market is witnessing a surge in collaborations between biotechnology companies, pharmaceutical firms, and academic research institutions. Such partnerships accelerate biomarker discovery, validation, and commercialization. These alliances are essential in bridging the gap between laboratory findings and real-world clinical application, helping bring advanced diagnostics closer to routine healthcare.
- Integration of AI and Digital Health: Artificial intelligence and digital platforms are being increasingly integrated into biomarker analysis. Machine learning algorithms can process vast amounts of biomarker data to detect subtle disease patterns, predict disease progression, and enable personalized treatment plans. This integration not only improves accuracy but also supports continuous, real-time monitoring of patients.
- Growing Role in Clinical Trials: Biomarkers have become indispensable in the drug development process. They are being used to identify suitable patients, track therapeutic responses, and evaluate drug safety more effectively. This trend reduces clinical trial timelines and costs while improving the chances of regulatory approval for new NASH therapies.
What Vitamin is Used in NASH?
Vitamin E is one of the most extensively studied supplements in the treatment of non-alcoholic steatohepatitis (NASH). Clinical evidence suggests that Vitamin E can help reduce oxidative stress, lower liver inflammation, and improve certain histological features of the liver in non-diabetic patients with NASH. Its antioxidant properties make it a promising therapeutic option, particularly for individuals who are not yet eligible for pharmacological treatments. However, Vitamin E is not a universal solution; some patients may not experience significant improvement, and long-term use may carry risks that require careful medical supervision. Because of this, healthcare providers typically recommend Vitamin E supplementation only after a thorough evaluation, often in combination with lifestyle modifications such as diet and exercise.
Regional Insights: U.S. Hotspots for NASH Biomarker Development
The U.S. remains a central hub for NASH biomarker innovation, with research and commercialization concentrated in regions with both high disease prevalence and strong biomedical infrastructure.
- Southeast and Midwest: These regions face some of the highest obesity and diabetes rates in the country, creating a significant patient base for NASH diagnosis and biomarker validation. Hospitals and regional healthcare systems are increasingly adopting non-invasive biomarker testing to manage the growing disease burden.
- Northeast (Boston, New York): Academic and medical research centers in this region are at the forefront of NASH biomarker discovery. With strong ties to pharmaceutical companies, the Northeast has become a leading hub for early-stage clinical trials, translational research, and biomarker standardization efforts.
- West Coast (California, San Diego, San Francisco): Known for its biotech ecosystem, the West Coast is driving innovation in digital health and AI-powered biomarker platforms. Collaborations between startups, academic institutions, and pharmaceutical firms are accelerating the commercialization of cutting-edge diagnostics.
- Texas and Southwest: With high liver disease prevalence and a growing number of specialty clinics, Texas is emerging as a strategic location for biomarker adoption and large-scale clinical trial recruitment.
U.S. Non-alcoholic Steatohepatitis (NASH) Biomarkers Market Dynamics
Drivers
The U.S. NASH biomarkers market is primarily driven by the rising burden of lifestyle-related diseases such as obesity, type 2 diabetes, and metabolic syndrome, which significantly increase the risk of liver disorders. Healthcare systems are also placing a stronger emphasis on preventive care, making early detection tools like biomarkers essential for reducing long-term healthcare costs. Additionally, the growing demand for personalized medicine is fueling adoption, as biomarkers enable physicians to tailor treatment strategies to individual patients, improving both clinical outcomes and overall treatment efficiency.
Opportunities
The market presents substantial opportunities for growth, particularly with the expansion of the therapeutic pipeline. As multiple NASH drugs move through development, biomarkers are expected to play a vital role in identifying suitable patients and tracking treatment effectiveness, creating vast potential for diagnostic companies. The commercialization of non-invasive solutions such as blood-based and imaging biomarkers is another key opportunity, offering safer and more convenient alternatives to liver biopsies. Furthermore, increased venture capital investments and government support for liver disease research are providing favorable conditions for biomarker innovation and broader market expansion.
Challenges
Despite positive momentum, the market faces several challenges that could slow adoption. A major hurdle is the lack of standardization, as variability in biomarker performance and the absence of universal testing protocols limit clinical reliability. Regulatory barriers also pose significant difficulties, with strict FDA requirements and lengthy approval processes delaying commercialization. High costs and limited reimbursement options remain another barrier, restricting patient access to advanced diagnostic tests. In addition, an awareness gap among certain healthcare providers continues to hinder widespread utilization, as not all physicians are familiar with the latest biomarker technologies.
Want custom data? Click here: https://www.visionresearchreports.com/report/customization/41348
Impact on the Healthcare Ecosystem
The widespread adoption of NASH biomarkers is set to create significant ripple effects across the U.S. healthcare system. For patients, these tools offer improved comfort, reduced risks, and greater trust in diagnostic procedures by replacing invasive liver biopsies with safer alternatives. Physicians gain access to real-time, evidence-based insights that enhance decision-making and improve treatment efficiency. Pharmaceutical companies benefit from faster and more efficient clinical trials, reduced development costs, and higher success rates for NASH drugs, while payers and insurers stand to achieve long-term cost savings through early detection and the prevention of advanced liver complications. Ultimately, the integration of biomarkers is not only transforming the way NASH is managed but also establishing a model for the future of diagnostics in other chronic diseases.
Case Study: Innovation Driving Market Growth
A leading U.S. biotechnology company, in collaboration with renowned academic research institutions, developed and validated a novel blood-based biomarker for the detection of NASH. Unlike traditional diagnostic methods, this biomarker demonstrated high accuracy in distinguishing NASH from simple fatty liver disease, while offering improved sensitivity and specificity that provided physicians with reliable diagnostic support. It also proved valuable in clinical trials, where it was successfully used to stratify patients and measure therapeutic responses more effectively. This collaboration not only accelerated the biomarker’s pathway toward FDA consideration but also attracted substantial investments from venture capital firms. The case underscores how strategic partnerships between industry and academia can fast-track innovation, strengthen investor confidence, and ultimately reshape the competitive landscape of the U.S. NASH biomarkers market.
Applications in the Market
Biomarkers are playing a transformative role in how non-alcoholic steatohepatitis (NASH) is diagnosed and managed across the U.S. healthcare system. They allow for early detection and screening, enabling physicians to identify the disease in its initial stages before symptoms appear, which makes timely intervention possible. Beyond diagnosis, biomarkers are also valuable in disease monitoring, as they provide real-time insights into whether a patient’s liver health is improving, stable, or deteriorating. By analyzing biomarker patterns, clinicians can predict how patients are likely to respond to therapy, which supports the growing shift toward personalized treatment strategies. In pharmaceutical research, biomarkers are critical to clinical trials, helping companies select the right patient groups, measure drug effectiveness more accurately, and reduce the time and cost of development. Most importantly, the adoption of non-invasive biomarker-based tools significantly reduces reliance on painful liver biopsies, improving patient compliance, comfort, and overall quality of care.
Read More: https://www.heathcareinsights.com/phosphoramidite-market/
U.S. Non-alcoholic Steatohepatitis Biomarkers Market Key Players
- GENFIT
- Prometheus Laboratories.
- Siemens Medical Solutions USA, Inc.
- Quest Diagnostics
- AstraZeneca
- Laboratory Corporation of America Holdings.
- Pfizer, Inc.
- Bristol-Myers Squibb Company
U.S. Non-alcoholic Steatohepatitis Biomarkers Market Segmentation
By Type
- Serum Biomarkers
- Hepatic Fibrosis Biomarkers
- Apoptosis Biomarkers
- Oxidative Stress Biomarkers
- Others
By End-use
- Pharma & CRO Industry
- Hospitals
- Diagnostic Labs
- Academic Research Institutes
By Region
- West
- Midwest
- Northeast
- Southwest
- Southeast
Future Outlook: What Lies Ahead
The future of the U.S. NASH biomarkers market appears highly promising, driven by a combination of technological, clinical, and policy advancements. Innovations such as next-generation sequencing, AI-powered analytics, and liquid biopsy technologies are expected to significantly improve the accuracy and accessibility of biomarker testing. Growing awareness among both physicians and patients will support mainstream adoption, making non-invasive biomarker tests a standard diagnostic tool in hospitals and clinics. As precision medicine gains momentum, biomarkers will play a central role in tailoring treatment plans and ensuring patients receive the most effective therapy. At the same time, increasing recognition of NASH as a major public health challenge is likely to prompt stronger regulatory and reimbursement support, further boosting adoption. By the end of the decade, biomarker-driven diagnosis and monitoring are projected to replace invasive liver biopsies as the gold standard in NASH management, transforming the way this disease is detected and treated.
Buy this Premium Research Report@ https://www.visionresearchreports.com/report/checkout/41348
You can place an order or ask any questions, please feel free to contact
sales@visionresearchreports.com| +1 650-460-3308
