The global lung cancer liquid biopsy market size was valued at approximately USD 1.11 billion in 2024 and is expected to reach nearly USD 3.43 billion by 2033, expanding at a compound annual growth rate (CAGR) of 13.35% between 2024 and 2033.
The market is driven by the rising prevalence of lung cancer worldwide and the growing demand for early detection and personalized treatment. Advancements in molecular diagnostics, increasing R&D investments, and the limitations of traditional tissue biopsies, such as invasiveness and difficulty in repeated testing are further fueling the adoption of liquid biopsy solutions.
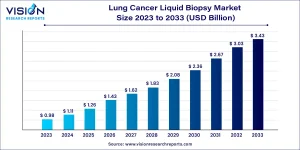
Lung Cancer Liquid Biopsy Market Overview
The Lung Cancer Liquid Biopsy Market is emerging as a transformative segment within the oncology diagnostics landscape. Liquid biopsy, a minimally invasive technique, enables the detection of cancer-related genetic material, such as circulating tumor DNA (ctDNA) and circulating tumor cells (CTCs), from blood or other bodily fluids. This approach offers a real-time assessment of tumor progression and treatment response, making it a highly valuable alternative to traditional tissue biopsies. The increasing incidence of lung cancer globally, coupled with advancements in molecular diagnostics, is accelerating the adoption of liquid biopsy technologies across hospitals, research institutions, and diagnostic laboratories.
Access Your Sample Report Today: https://www.visionresearchreports.com/report/sample/41333
Lung Cancer Liquid Biopsy Market Growth
The lung cancer liquid biopsy market has been witnessing robust growth due to the growing emphasis on personalized medicine and precision oncology. Clinicians are increasingly relying on liquid biopsies for early detection, disease monitoring, and therapeutic guidance. Integration of advanced molecular profiling techniques and next-generation sequencing (NGS) has further enhanced the accuracy and reliability of liquid biopsy tests, attracting investments from biotechnology companies and research institutions.
Additionally, the increasing awareness of non-invasive diagnostic methods among patients and healthcare providers is driving higher market penetration. Collaborations between biotech firms and pharmaceutical companies for companion diagnostics and targeted therapies are expected to propel market expansion. Continuous innovation in biomarker discovery and assay development is anticipated to support long-term growth while addressing the limitations of conventional biopsy procedures.
Lung Cancer Liquid Biopsy Market Trends
- Rising Adoption of Next-Generation Sequencing (NGS): NGS technologies are increasingly used to detect rare mutations in liquid biopsies, enabling more precise early-stage diagnosis and better-informed therapy selection. This trend is driving innovation in test accuracy and reliability.
- Integration with Personalized Medicine: Liquid biopsies allow clinicians to tailor treatment plans based on each patient’s tumor profile, enhancing therapeutic outcomes and reducing unnecessary interventions. Personalized medicine is becoming a standard approach in lung cancer management.
- Emergence of Multi-Cancer Panels: Companies are developing advanced liquid biopsy panels capable of detecting multiple types of cancer in a single test, expanding the clinical potential beyond lung cancer and supporting preventive oncology initiatives.
- Focus on Companion Diagnostics: Collaborative efforts between diagnostics and pharmaceutical companies are increasing the availability of companion diagnostics, which help match patients with targeted therapies more efficiently and safely.
- Expansion of Non-Invasive Monitoring: Liquid biopsies are being adopted for ongoing monitoring of treatment response and disease progression, reducing the need for repeated invasive tissue biopsies and improving patient comfort.
- Integration of AI and Bioinformatics: Artificial intelligence and advanced bioinformatics tools are being integrated with liquid biopsy data to improve mutation detection, predict therapy response, and streamline clinical decision-making.
- Growing Use in Clinical Trials: Pharmaceutical companies are increasingly using liquid biopsies in clinical trials to monitor genetic changes, track treatment efficacy, and accelerate drug development for lung cancer therapies.
Lung Cancer Liquid Biopsy Market Dynamics
Drivers
The lung cancer liquid biopsy market is primarily driven by the rising prevalence of lung cancer worldwide and the growing demand for early detection and personalized treatment approaches. The limitations of traditional tissue biopsies, including invasiveness and difficulty in repeated sampling, further emphasize the need for liquid biopsy solutions. Additionally, technological advancements in molecular diagnostics, coupled with increasing R&D investments, are fueling market growth.
Opportunities
Expanding applications of liquid biopsy in monitoring treatment resistance, detecting minimal residual disease, and enabling early-stage cancer detection present significant opportunities. Emerging markets with growing healthcare infrastructure and increasing awareness about non-invasive diagnostic solutions are untapped avenues for growth. Partnerships with biotech startups focusing on novel biomarker discovery also offer opportunities for market players.
Challenges
High costs associated with liquid biopsy tests, regulatory hurdles, and lack of standardization in assay protocols pose challenges for widespread adoption. Furthermore, the sensitivity and specificity of certain liquid biopsy tests may vary depending on tumor type and stage, requiring continuous validation and improvement.
Want custom data? Click here: https://www.visionresearchreports.com/report/customization/41333
What Is the Role of Liquid Biopsy in Lung Cancer?
Liquid biopsy plays a vital role in the management of lung cancer by offering a minimally invasive way to gain critical insights into tumor behavior. It enables early detection by identifying tumor-derived genetic alterations before symptoms arise, allowing timely intervention. Throughout treatment, clinicians can use liquid biopsy for real-time monitoring of how a patient’s tumor is responding, making it easier to adjust therapies as needed. It is also effective in detecting resistance mutations, helping guide the selection of next-line treatments when tumors become resistant to targeted therapies. After surgery or therapy, liquid biopsies can identify minimal residual disease (MRD), detecting traces of remaining cancer cells to prevent relapse. Overall, as a non-invasive alternative to traditional tissue biopsies, liquid biopsy reduces patient risk and allows repeated testing, making it a highly valuable tool in modern lung cancer care.
What Is the Difference Between a Liquid Biopsy and a Traditional Tissue Biopsy?
- Procedure: A liquid biopsy involves a simple blood draw or collection of other bodily fluids, making it minimally invasive. In contrast, a traditional tissue biopsy requires surgical or needle-based extraction of tumor tissue, which is invasive and can be risky for patients.
- Frequency: Liquid biopsies can be performed multiple times, allowing for ongoing monitoring of tumor progression. Traditional tissue biopsies, however, are limited in frequency due to their invasive nature and associated patient risk.
- Detection: Liquid biopsy identifies circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and other biomarkers in the bloodstream. Traditional biopsies detect tumor cells directly from the tissue sample, providing a snapshot of the tumor at a specific location.
- Speed: Liquid biopsies provide faster results and are suitable for real-time monitoring of disease progression and treatment response. Traditional tissue biopsies take longer, as they require processing and pathological examination.
- Application: Liquid biopsy is increasingly used for early detection, treatment monitoring, and identification of resistance mutations. Traditional biopsies remain critical for initial diagnosis, tumor characterization, and staging.
Regional Analysis of the Lung Cancer Liquid Biopsy Market
- North America
North America remains the largest market for lung cancer liquid biopsy, driven by its well-established healthcare infrastructure, high adoption of advanced diagnostic technologies, and strong research and development activities. The presence of leading biotechnology companies and frequent clinical trials in the U.S. and Canada further bolster market growth. Increasing awareness among patients and healthcare providers about the benefits of non-invasive diagnostics is also accelerating adoption in this region.
- Europe
Europe is a significant market, supported by favorable government initiatives, reimbursement frameworks, and strong clinical research networks. Countries such as Germany, the U.K., and France are witnessing high adoption rates due to proactive healthcare policies and increasing investment in precision medicine. The region also benefits from a growing focus on personalized oncology treatments and collaborations between pharmaceutical and diagnostic companies.
- Asia-Pacific
The Asia-Pacific market is emerging as a high-growth region due to the rising prevalence of lung cancer, expanding healthcare infrastructure, and growing awareness of innovative diagnostic methods. Countries such as China, Japan, and India are investing heavily in molecular diagnostics and liquid biopsy technologies. The availability of cost-effective diagnostic solutions and increasing healthcare expenditure are expected to drive further market penetration in this region.
- Latin America
Latin America is witnessing gradual adoption of liquid biopsy technologies, driven by improvements in healthcare facilities, increasing patient awareness, and expanding oncology services. Countries like Brazil and Mexico are seeing increased investments in molecular diagnostics and precision oncology, providing opportunities for market growth.
- Middle East & Africa
The Middle East & Africa region is still developing its liquid biopsy market. Growth is fueled by rising healthcare expenditure, increasing cancer awareness campaigns, and the introduction of innovative diagnostic technologies in urban healthcare centers. Countries such as Saudi Arabia and the UAE are gradually adopting advanced oncology diagnostics, paving the way for future market expansion.
What Are the Applications of Liquid Biopsy in the Clinic?
Liquid biopsy has a wide range of applications in the clinical management of lung cancer. It plays a key role in personalized medicine by identifying specific mutations that allow clinicians to select targeted therapies tailored to an individual’s tumor profile. It is also used for treatment monitoring, tracking changes in tumor genetics to assess therapy effectiveness and detect resistance at an early stage. Liquid biopsy supports screening of high-risk patients, particularly smokers or individuals with a family history of lung cancer, enabling earlier intervention. In addition, it aids companion diagnostics, helping pharmaceutical companies develop drugs and match patients with suitable treatments, and is increasingly used in clinical trials as a non-invasive method to monitor patient response and improve trial efficiency. Its versatility and minimally invasive nature make liquid biopsy an invaluable tool for oncologists, researchers, and pharmaceutical companies alike.
Case Study: Transforming Lung Cancer Care with Liquid Biopsy
A leading biotechnology company partnered with top oncology hospitals to launch a comprehensive liquid biopsy program aimed at monitoring lung cancer progression in patients receiving targeted therapies. The program leveraged next-generation sequencing (NGS) to analyze circulating tumor DNA (ctDNA) and detect genetic mutations associated with therapy resistance in real-time.
Through this initiative, clinicians were able to adjust treatment plans promptly, switching therapies when resistance mutations were detected. This proactive approach not only improved patient survival rates but also reduced unnecessary exposure to ineffective treatments, demonstrating both clinical and economic value.
Read More: https://www.heathcareinsights.com/oligonucleotide-synthesis-market/
Lung Cancer Liquid Biopsy Market Key Players
- Eurofins Scientific
- MDxHealth
- CareDx
- Immucor
- Thermo Fisher Scientific Inc.
- Menarini Silicon Biosystems
- Qiagen
- Guardant Health
- Exact Sciences Corporation
- Myriad Genetics, Inc.
- LungLife AI, Inc.
- Bio-Rad Laboratories
- Agilent Technologies
Lung Cancer Liquid Biopsy Market Segmentation
By Sample Type
- Blood Sample Based
- Others
By Biomarker
- Circulating Nucleic Acids
- CTC
- Exosomes/Microvesicles
- Circulating Proteins
By Technology
- Multi-gene-parallel Analysis (NGS)
- Single Gene Analysis (PCR Microarrays)
By End-use
- Hospitals & Laboratories
- Specialty Clinics
- Academic & Research Centers
- Others
By Clinical Application
Therapy Selection
Treatment Monitoring
Early Cancer Screening
Recurrence Monitoring
Others
By Product
- Instruments
- Consumables Kits and Reagents
- Software and Services
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa (MEA)
Future Outlook
The future of the lung cancer liquid biopsy market looks promising, with innovations in multi-gene panels, AI-driven diagnostics, and real-time disease monitoring expected to enhance adoption. Increasing integration of liquid biopsies in clinical trials and routine healthcare settings will likely improve treatment outcomes and reduce the burden of invasive procedures. Furthermore, ongoing research in liquid biopsy biomarkers and companion diagnostics will continue to expand its clinical relevance across oncology.
Buy this Premium Research Report@ https://www.visionresearchreports.com/report/checkout/41333
You can place an order or ask any questions, please feel free to contact
sales@visionresearchreports.com| +1 650-460-3308
