The Asia Pacific biopharma excipients market is shaped by a diverse mix of rapidly growing economies and mature pharmaceutical hubs. Each country in the region contributes uniquely to market expansion, driven by local innovation, government support, and increasing global integration. From manufacturing powerhouses to regulatory leaders, these markets are collectively fueling the region’s rise as a global center for excipient development and supply.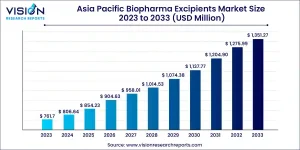
Asia Pacific Biopharma Excipients Market Overview
The Asia Pacific region is emerging as a key player in the global biopharmaceutical industry, with a growing focus on innovative drug development and advanced formulations. Excipients, which are essential non-active ingredients used in drug production, are gaining importance as they help improve drug stability, absorption, and patient compliance. As biopharma companies in the region expand their capabilities, the demand for high-quality, functional excipients is steadily rising.
The increasing production of biologics, including monoclonal antibodies, vaccines, and gene therapies, is fueling the need for specialized excipients. These advanced therapies require precise formulation support to maintain effectiveness throughout manufacturing, storage, and delivery. Local governments across Asia Pacific are also supporting pharmaceutical growth through favorable regulations, infrastructure development, and funding for research and development.
Get a Sample@ https://www.visionresearchreports.com/report/sample/41628
Asia Pacific Biopharma Excipients Market Growth Fueling Innovation and Quality
One of the primary growth drivers of the Asia Pacific biopharma excipients market is the increasing emphasis on biologics and biosimilars. As the region becomes a major hub for pharmaceutical manufacturing and R&D, especially in countries like India, China, South Korea, and Japan, the demand for specialized excipients to stabilize, preserve, and deliver biologic drugs efficiently is escalating. Biopharmaceutical companies are increasingly investing in the development of complex formulations that rely heavily on functional and safe excipients.
Moreover, the evolving regulatory landscape across Asia Pacific is boosting the demand for compliant and high-purity excipients. Regulatory bodies such as the CDSCO (India) and NMPA (China) are aligning with global standards, encouraging local manufacturers to elevate formulation quality. This transition is prompting excipient suppliers to innovate and customize their products for emerging biopharma needs, further fueling the market’s growth.
Market Trends Shaping the Future of Biopharma Excipients
- Rise of Injectable Biologics: There is a growing demand for excipients that enhance the stability and shelf life of injectable formulations, particularly for monoclonal antibodies and mRNA-based therapies.
- Focus on Biocompatibility and Safety: Manufacturers are prioritizing excipients that offer low immunogenicity, improved bioavailability, and patient safety, especially for sensitive therapeutic areas like oncology and autoimmune disorders.
- Expansion of Local Manufacturing Capacities: Countries like India and China are ramping up domestic excipient production to reduce dependency on imports and meet the needs of local biopharma players.
Asia Pacific Biopharma Excipients Market Dynamics
Drivers
The biopharma excipients market in Asia Pacific is being propelled by the rapid growth in biologics and biosimilars manufacturing. As pharmaceutical companies scale up production of complex biologic therapies, the demand for reliable and functional excipients is becoming increasingly critical to ensure product stability, safety, and performance.
Rising healthcare needs, driven by a growing and aging population across the region, are also contributing to market expansion. As more patients seek advanced treatments for chronic and lifestyle-related diseases, the demand for biologics and the excipients that support their formulation is on the rise.
In parallel, technological advancements in excipient development are opening up new possibilities for drug delivery and patient-centric formulations. Innovations such as multifunctional excipients, smart polymers, and nanotechnology-based carriers are enabling more effective and targeted therapies, boosting the overall value of excipients in modern drug development.
Opportunities
There is significant opportunity in expanding into niche therapeutic areas such as oncology, autoimmune disorders, and rare diseases. These segments often require specialized formulations and delivery mechanisms, creating a strong need for high-performance excipients tailored to specific medical needs.
Strategic collaboration between local excipient manufacturers and global pharmaceutical companies is fostering knowledge transfer, enhancing innovation, and accelerating market entry. These partnerships are helping local players access advanced technologies and regulatory insights while offering global companies a cost-effective manufacturing base.
Challenges
One of the biggest challenges facing the market is the lack of harmonized regulatory frameworks across Asian countries. Each country often has its own standards and approval processes, making it difficult for companies to scale formulations across borders without facing delays or compliance issues.
The high cost of manufacturing high-purity or specialty excipients also presents a barrier, particularly for smaller players and startups. These advanced excipients require stringent quality control and specialized production environments, adding to the financial burden.
Market Growth Fueling Innovation and Quality
One of the primary growth drivers of the Asia Pacific biopharma excipients market is the increasing emphasis on biologics and biosimilars. As the region becomes a major hub for pharmaceutical manufacturing and R&D, especially in countries like India, China, South Korea, and Japan, the demand for specialized excipients to stabilize, preserve, and deliver biologic drugs efficiently is escalating. Biopharmaceutical companies are increasingly investing in the development of complex formulations that rely heavily on functional and safe excipients.
Moreover, the evolving regulatory landscape across Asia Pacific is boosting the demand for compliant and high-purity excipients. Regulatory bodies such as the CDSCO (India) and NMPA (China) are aligning with global standards, encouraging local manufacturers to elevate formulation quality. This transition is prompting excipient suppliers to innovate and customize their products for emerging biopharma needs, further fueling the market’s growth.
Market Trends Shaping the Future of Biopharma Excipients
- Rise of Injectable Biologics: There is a growing demand for excipients that enhance the stability and shelf life of injectable formulations, particularly for monoclonal antibodies and mRNA-based therapies.
- Focus on Biocompatibility and Safety: Manufacturers are prioritizing excipients that offer low immunogenicity, improved bioavailability, and patient safety, especially for sensitive therapeutic areas like oncology and autoimmune disorders.
- Expansion of Local Manufacturing Capacities: Countries like India and China are ramping up domestic excipient production to reduce dependency on imports and meet the needs of local biopharma players.
- Integration of Nanotechnology in Excipients: Nanocarrier-based excipients are being explored for targeted drug delivery, especially in cancer treatment and advanced biologic formulations.
What’s Driving the Biopharma Excipients Boom in Asia?
The biopharma industry in Asia is experiencing rapid growth, and excipients are playing a critical role in this transformation. Several key factors are fueling the demand for biopharma excipients across the region. First, there’s a noticeable increase in the development and manufacturing of biologics, such as monoclonal antibodies, vaccines, and biosimilars. These complex drugs require high-quality, specialized excipients to ensure stability and effectiveness.
Second, countries like China, India, South Korea, and Japan are expanding their pharmaceutical infrastructure and investing heavily in R&D. Government initiatives and favorable policies aimed at strengthening local production capabilities are encouraging the use of advanced excipients in drug formulations. Additionally, growing awareness about regulatory compliance and quality standards is driving the need for reliable excipient sources.
Top Trends Shaping the Future of Excipients
- Rise in Demand for Injectable Formulations: Injectable biologics require excipients that enhance solubility, stability, and bioavailability, creating a surge in demand for functional excipients.
- Increased Focus on Patient-Centric Formulations: Excipients are being designed to improve the patient experience, including taste-masking agents, rapid-release polymers, and non-allergenic stabilizers.
- Adoption of Novel Drug Delivery Technologies: Nanotechnology and liposomal excipients are being explored for targeted delivery in treatments like cancer and gene therapy.
- Shift Towards Sustainable and Natural Excipients: Manufacturers are increasingly developing eco-friendly and plant-based excipients to meet environmental and consumer expectations.
What Are the Main Types of Excipients?
Biopharma excipients come in various forms, each serving a specific purpose in drug formulation. Some of the major types include:
- Polymers – Used as binders, controlled-release agents, and stabilizers
- Alcohols and Polyols – Help in improving solubility and act as preservatives
- Sugars and Amino Acids – Serve as cryoprotectants in biologics and vaccines
- Surfactants – Aid in emulsification and protein stabilization
- Inorganic Salts – Maintain pH balance and tonicity in injectable drugs
These excipients are carefully selected based on the formulation needs and route of administration—whether it’s oral, injectable, or topical.
Where Are Excipients Used in Biopharma?
Excipients are essential in a wide range of biopharmaceutical applications. They are not just “inactive” ingredients but play an active role in ensuring the effectiveness, safety, and patient acceptability of the final product. Key application areas include:
- Monoclonal Antibodies (mAbs) – Require stabilizers to maintain protein structure
- Vaccines – Need preservatives and adjuvants for stability and immune response
- mRNA and Gene Therapies – Utilize lipid-based excipients for targeted delivery
- Controlled Drug Delivery – Use polymers for timed release of medication
Topical and Oral Biologics – Involve emulsifiers and taste-masking agents for better user experience
Applications in the Market
Biopharma excipients are essential components in a wide range of drug formulations, serving functions that go far beyond simple fillers or binders. One of their key applications is in the production of monoclonal antibodies (mAbs), where excipients help stabilize proteins, prevent aggregation, and extend shelf life. In vaccine formulations, including mRNA and viral vector-based vaccines, excipients play a crucial role in maintaining the integrity of active ingredients, enhancing immune response, and ensuring safe delivery.
In gene and cell therapy products, which represent some of the most cutting-edge advances in modern medicine, specialized excipients are used to protect genetic material, improve delivery mechanisms, and ensure bioavailability. The development of biosimilars and biobetters which aim to replicate or enhance existing biologic therapies—also heavily depends on excipients for maintaining equivalency and performance standards.
Case Study: India’s Role in Scaling Biologic Formulations
India has emerged as a significant contributor to the Asia Pacific biopharma excipients market. A leading Indian pharmaceutical company partnered with a European excipient supplier to develop a temperature-stable injectable biologic. By using proprietary sugar-based stabilizers, the company was able to extend the product’s shelf life and gain swift regulatory approval in multiple emerging markets. This collaboration underscores the growing trend of international partnerships and local innovation in the excipients space.
Read More: https://www.visionresearchreports.com/cellulose-fiber-market/41629
Top Manufactures in Asia Pacific Biopharma Excipients Market
- Signet Excipients Pvt. Ltd.
- ABITEC
- Sigachi Industries.
- Roquette Frères
- Colorcon
- Meggle GmbH & Co. KG
- CLARIANT
- DFE Pharma
- SPI Pharma
- IMCD.
- Spectrum Chemical
- Pharmonix
- BASF SE
Want custom data? Click here: https://www.visionresearchreports.com/report/customization/41628
Asia Pacific Biopharma Excipients Market Segmentation
By Product
- Solubilizers & Surfactants/Emulsifiers
- Triglycerides
- Esters
- Others
- Polyols
- Mannitol
- Sorbitol
- Others
- Carbohydrates
- Sucrose
- Dextrose
- Starch
- Others
- Specialty Biopharma Excipients/Others
Regional Analysis
The Asia Pacific region offers a dynamic and evolving landscape for the biopharma excipients market. Each country contributes uniquely to the growth of the sector, backed by varying levels of industrial maturity, regulatory focus, and innovation capabilities. Here’s a closer look at the major contributors:
- China
China stands at the forefront of the regional biopharma excipients market, both in terms of production volume and technological advancement. The Chinese government continues to pour resources into building biotech innovation zones, expanding pharmaceutical industrial parks, and supporting high-end manufacturing. These initiatives are strengthening domestic excipient production and improving access to global supply chains.
China is also becoming a leader in advanced formulations, thanks to its rapid adoption of biologics, biosimilars, and novel drug delivery systems. Local companies are forming strategic partnerships with international firms, enabling knowledge transfer and improving compliance with global regulatory standards such as ICH and US FDA.
- India
India is one of the fastest-growing markets for pharmaceutical manufacturing, particularly known for its dominance in generic drugs and biosimilars. This growth directly supports the rising demand for excipients tailored to these formulations. With a well-established infrastructure of API and excipient manufacturers, India is rapidly expanding its presence in both domestic and international biopharma markets.
Government-backed initiatives like “Make in India” and PLI (Production-Linked Incentive) Schemes are encouraging large-scale pharmaceutical and excipient production. Additionally, Indian excipient manufacturers are increasingly investing in R&D, quality control systems, and international certifications to cater to regulated markets in the US, Europe, and Southeast Asia.
- Japan
Japan represents a mature and quality-driven pharmaceutical market with a strong focus on innovation and precision. The country’s strict regulatory framework encourages high levels of compliance, making it a hub for premium-grade excipients. Japanese companies often lead in the development of sophisticated drug delivery systems, including sustained-release and targeted therapies.
Given the aging population in Japan, there is a significant demand for elderly-friendly dosage forms. As a result, excipients that improve drug solubility, bioavailability, and patient adherence are in high demand. Additionally, Japan is exploring natural and biodegradable excipients as part of its commitment to sustainable healthcare.
- South Korea
South Korea is rapidly emerging as a biotechnology and pharmaceutical innovation hub in Asia. Supported by strong government policies, the country is actively promoting public-private partnerships, investing in biotech clusters, and enhancing its excipient manufacturing infrastructure.
South Korea’s emphasis on innovation extends into biopharma excipients, with a focus on excipients for cell and gene therapy, mRNA vaccines, and high-value biologics. Companies are developing capabilities in high-purity excipient production, particularly those used in parenteral and injectable formulations.
Future Outlook
The future of the Asia Pacific biopharma excipients market looks promising, driven by the ongoing expansion of biologic drug pipelines, increasing investments in pharmaceutical infrastructure, and the rapid adoption of advanced drug delivery technologies. Customized excipients designed for specific therapies such as oncology, immunology, and rare diseases will gain traction. Furthermore, as the region continues to embrace global regulatory harmonization and quality standards, opportunities for global players and domestic innovators will multiply.
Buy this Premium Research Report@https://www.visionresearchreports.com/report/checkout/41628
You can place an order or ask any questions, please feel free to contact
sales@visionresearchreports.com| +1 650-460-3308
